| Isotope | Atomic mass (Da) | Isotopic abundance (amount fraction) |
|---|---|---|
| 63Cu | 62.929 597(3) | 0.6915(15) |
| 65Cu | 64.927 790(5) | 0.3085(15) |
Copper is a reddish orange metal that has the atomic number 29 in the periodic table. It is a Transition metal and located in Group 11 of the periodic table. It has the symbol Cu. General, Atomic and Crystallographic Properties and Features of Copper. Source: 'Properties of Copper and Copper Alloys at Cryogenic Temperatures' by N.J. Drexler, and R.P. Reed ( NIST MN 177) General and Atomic Properties of Copper.

In 1961, the Commission recommended Ar(Cu) = 63.54 based on the recent chemical determinations. In 1967, the Commission recommended Ar(Cu) = 63.546(1) based on the mass-spectrometric measurements. The recommended uncertainty was increased to 0.003 in 1969 to include natural variations of up to approx. 0.15 % in the isotopic abundances of copperisotopes, and given the annotation 'r' to indicate that the precision was limitedby natural variability.
In the compilation by the Commission, the lowest reported δ65Cu value in a naturallyoccurring sample is −7.65 ‰ (x(65Cu) = 0.3066 and Ar(Cu) = 63.542) for a specimenof a Cu-chloride mineral (atacamite) from Chile. The highest reported δ65Cu is +9 ‰ (x(65Cu) = 0.3102 and Ar(Cu) = 63.549) for a Cu-carbonate mineral (aurichalcite) from Arizona. Some of these values are outside the range of the stated atomic-weight uncertainty andmay justify a re-evaluation by the Commission of the atomic-weight uncertainty or annotations
Atomic No Of Copper
© IUPAC 2003
CIAAW
Copper
Ar(Cu) = 63.546(3) since 1969
The name derives from the Latin cuprum for Cyprus, the island where the Romans first obtained copper.The symbol Cu also comes from the Latin cuprum. The element has been known since prehistoric times.
Ar(Cu) = 63.546(3) since 1969
The name derives from the Latin cuprum for Cyprus, the island where the Romans first obtained copper.The symbol Cu also comes from the Latin cuprum. The element has been known since prehistoric times.
Atomic No Of Copper Element
Isotopic reference materials of copper.
The atomic mass of an element is the average mass of the atoms of an element measured in atomic mass unit (amu, also known as daltons, D). The atomic mass is a weighted average of all of the isotopes of that element, in which the mass of each isotope is multiplied by the abundance of that particular isotope. (Atomic mass is also referred to as atomic weight, but the term 'mass' is more accurate.)
Atomic Number 36 is belong to element of Krypton. Chemical symbol for Krypton is Kr. Number of protons in Krypton is 36. Atomic weight of Krypton is 83.798 u or g/mol. 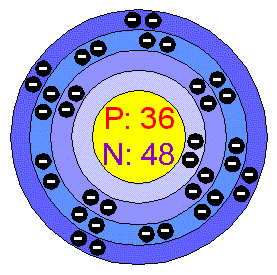

For instance, it can be determined experimentally that neon consists of three isotopes: neon-20 (with 10 protons and 10 neutrons in its nucleus) with a mass of 19.992 amu and an abundance of 90.48%, neon-21 (with 10 protons and 11 neutrons) with a mass of 20.994 amu and an abundance of 0.27%, and neon-22 (with 10 protons and 12 neutrons) with a mass of 21.991 amu and an abundance of 9.25%. The average atomic mass of neon is thus:
| 0.9048 | × | 19.992 amu | = | 18.09 amu |
| 0.0027 | × | 20.994 amu | = | 0.057 amu |
| 0.0925 | × | 21.991 amu | = | 2.03 amu |
| 20.18 amu |

The atomic mass is useful in chemistry when it is paired with the mole concept: the atomic mass of an element, measured in amu, is the same as the mass in grams of one mole of an element. Thus, since the atomic mass of iron is 55.847 amu, one mole of iron atoms would weigh 55.847 grams. The same concept can be extended to ionic compounds and molecules. One formula unit of sodium chloride (NaCl) would weigh 58.44 amu (22.98977 amu for Na + 35.453 amu for Cl), so a mole of sodium chloride would weigh 58.44 grams. Dropbox folder mac. One molecule of water (H2O) would weigh 18.02 amu (2×1.00797 amu for H + 15.9994 amu for O), and a mole of water molecules would weigh 18.02 grams.
The original periodic table of the elements published by Dimitri Mendeleev in 1869 arranged the elements that were known at the time in order of increasing atomic weight, since this was prior to the discovery of the nucleus and the interior structure of the atom. The modern periodic table is arranged in order of increasing atomic number instead. Markdown file online download.
